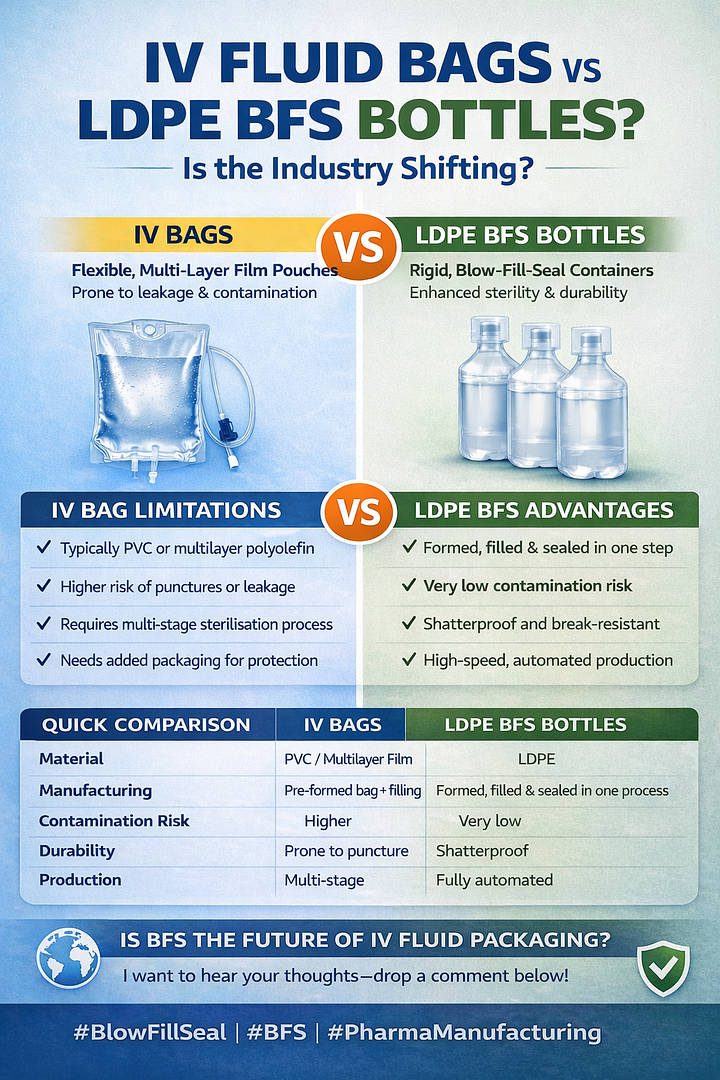
The pharmaceutical industry has traditionally relied on IV fluid bags made from PVC or multilayer polymer films to package large volume parenterals. These flexible bags are widely used across hospitals and healthcare facilities around the world.
However, as pharmaceutical manufacturing standards evolve and the demand for higher sterility assurance and automated production increases, many manufacturers are turning to Blow Fill Seal (BFS) technology using LDPE bottles.
So how do traditional IV fluid bags compare to LDPE Blow Fill Seal bottles, and why are more pharmaceutical companies adopting BFS packaging?
What Are IV Fluid Bags?
IV fluid bags are flexible pouches typically manufactured from PVC or multilayer polyolefin films. These bags are produced using form-fill-seal or bag-forming processes before being filled with sterile solutions.
They are commonly used for:
- Saline solutions
- Glucose solutions
- Electrolyte infusions
- Large volume parenterals (LVPs)
While IV bags have been the industry standard for decades, they present several challenges related to durability, sterility and transport.
Limitations of Traditional IV Bags
Although widely used, IV bags can present several risks during manufacturing, distribution and hospital handling.
Leakage and Puncture Risk
Flexible IV bags are more vulnerable to punctures and leakage during transport or storage, especially when handled in large volumes.
Contamination Risks
Traditional bag manufacturing often requires multiple production and sterilisation stages, increasing the risk of contamination compared to closed aseptic systems.
Additional Packaging Requirements
Because IV bags are flexible and more fragile, they typically require additional secondary packaging to protect them during shipping and storage.
These limitations have led pharmaceutical manufacturers to explore more robust sterile packaging technologies.
What Are LDPE Blow Fill Seal Bottles?
Blow Fill Seal (BFS) technology is an advanced aseptic manufacturing process that forms, fills and seals containers in one continuous automated operation.
Using low-density polyethylene (LDPE), BFS machines create sterile containers directly from polymer resin and immediately fill them with pharmaceutical liquid.
The entire process takes place within a closed sterile environment, reducing human interaction and contamination risk.
BFS packaging is widely used for:
- IV fluids
- Respiratory solutions
- Ophthalmic products
- Inhalation therapies
- Unit-dose pharmaceutical liquids
Advantages of LDPE Blow Fill Seal Bottles
Superior Sterility
One of the biggest advantages of BFS technology is the integrated aseptic process.
The container is:
- Formed from molten polymer
- Filled with sterile solution
- Sealed immediately
All within seconds and without human intervention.
This dramatically reduces contamination risk compared to traditional filling methods.
Reduced Contamination Risk
Because BFS manufacturing occurs inside an enclosed sterile chamber, it eliminates many of the contamination risks associated with multi-stage filling processes.
Minimal human contact makes BFS one of the most trusted aseptic technologies in pharmaceutical manufacturing.
Greater Durability
LDPE BFS bottles are shatterproof and break-resistant.
They are rigid enough to maintain shape during transport but flexible enough to allow controlled dispensing.
This durability reduces the likelihood of:
- Leakage
- Punctures
- Product loss
High-Efficiency Manufacturing
BFS systems are designed for high-volume automated production, allowing pharmaceutical manufacturers to produce large quantities of sterile liquid containers quickly and consistently.
This makes BFS an attractive option for manufacturers producing large volume parenterals (LVPs).
Why Blow Fill Seal Technology Is Growing in Pharmaceutical Packaging
The global pharmaceutical industry is increasingly adopting Blow Fill Seal technology due to its ability to deliver:
- Higher sterility assurance
- Reduced contamination risk
- Improved production efficiency
- Durable, tamper-resistant containers
For applications such as IV fluids, respiratory solutions, ophthalmics and biologics, BFS packaging offers a reliable alternative to traditional IV bags.
As pharmaceutical manufacturers continue to prioritise aseptic processing, automation and product safety, BFS technology is expected to play an even larger role in the future of sterile packaging.
Looking for Blow Fill Seal Equipment?
At GlobePack Equipment, we specialise in sourcing and supplying used and refurbished Blow Fill Seal machines for pharmaceutical manufacturers worldwide.
Our inventory regularly includes equipment from leading OEMs such as:
- Rommelag
- Weiler Engineering
- Brevetti Angela
If you are looking to expand your BFS production capacity, our team can help you find the right solution without the long lead times associated with new equipment.
👉 Contact us today to discuss available Blow Fill Seal machinery.